Advanced Ozone Technology for Cleaner, Safer Water
Municipal Water Treatment
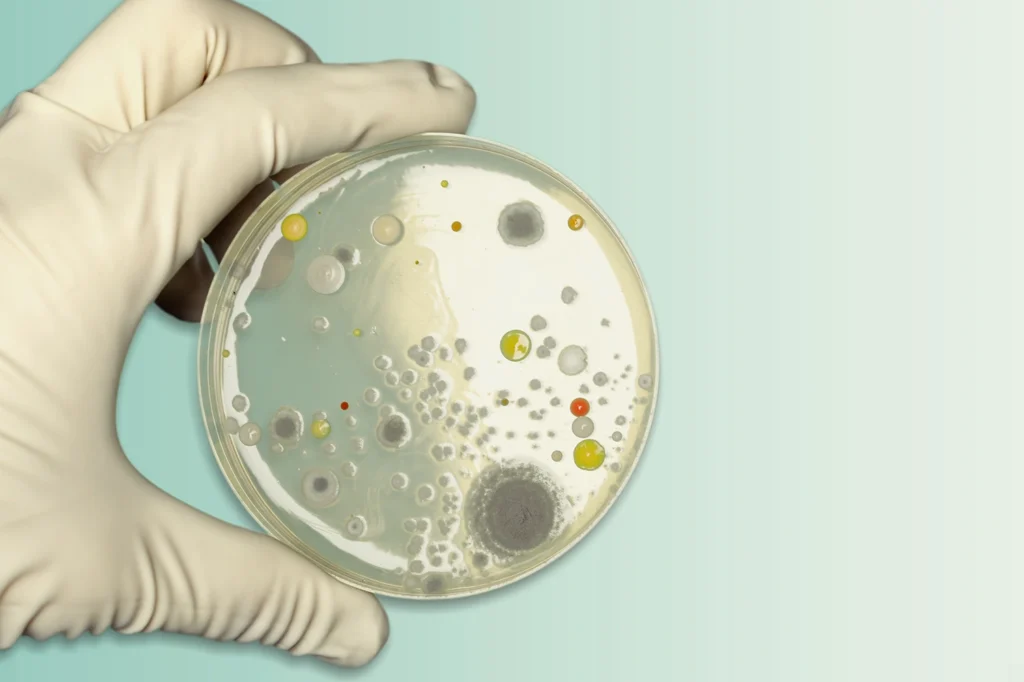
Clean, safe water plays an important role in maintaining public health and environmental sustainability. Our cutting-edge ozone technology offers a powerful and efficient solution for municipal water treatment facilities looking to improve water quality, overcome existing challenges, and meet regulatory standards. Purifico Ozone offers a range of ozone generators and related equipment, specifically designed for municipal water treatment applications. Our team of experts provides comprehensive support, from system design and installation to maintenance and training.