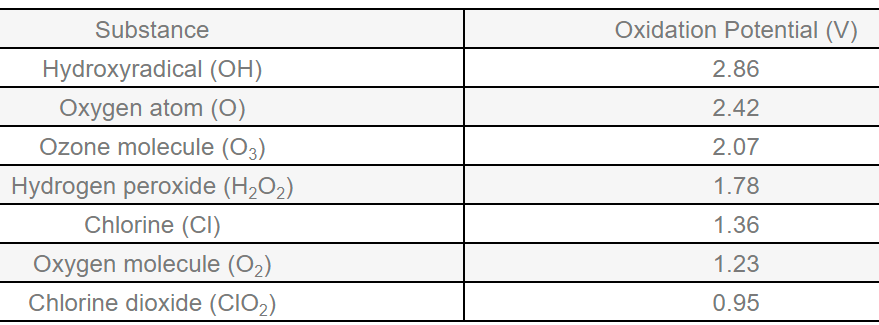
In some of the examples below, addition of oxygen to molecules is defined as oxidation. Over time, the definition of oxidation was broadened to include any reaction where a chemical loses an electron in a reaction (whether or not oxygen is involved). Every atom has a nucleus, and electrons that circle around it in ‘orbitals’, similar to how the planets orbit around the sun. Electrons in the outermost shell are referred to as ‘valence’ electrons. The number of valence electrons held by an atom affects how it interacts with other atoms. In turn, this affects how molecules form and the chemical reactions that occur between them. Oxidation occurs when a valence electron is removed. The removal of electrons through oxidation breaks bonds between atoms and re-arranges molecules in a destructive manner. These oxidative reactions make ozone useful for waterborne pathogen control and water disinfection, since it breaks down pathogens or pollutants.
• Iron that is exposed to O2 and H2O in the air will slowly rust, forming iron oxide.
4 Fe + 3 O2 + 6H2O → 4Fe(OH)3
• Carbon dioxide (CO2) is an oxide of carbon (C). It can be produced many ways, such as cellular respiration – the air you exhale when you breathe.
C6H12O6 + 6O2 → 6CO2 + 6H2O
• Water (H2O) is an oxide of hydrogen (H+). It can be produced in many ways, for example through combination of hydrogen and oxygen gasses
2H2 + O2 → 2H2O
When ozone accepts an electron (e–), oxidizing a target, it produces oxygen and hydroxyls. These by-products do not accumulate in solution and are not harmful to plants or livestock.
O3 + H2O + 2e– → O2 + 2 OH–
As shown below, when chlorine and chlorine dioxide are used for water treatment, they leave chloride residues behind in water. Chlorides are toxic to plants or livestock above certain concentrations and they accumulate in solution.
• When chlorine accepts an electron (e–), oxidizing a target, it produces chlorides.
Cl2 + 2e– → 2Cl–
• When chlorine dioxide accepts an electron (e–), oxidizing a target, it produces chlorites. Chlorites then undergo reactions that produce chlorides.
ClO2 + e– → ClO2–
ClO2– + 2H2O + 4e– → Cl– + 4OH–